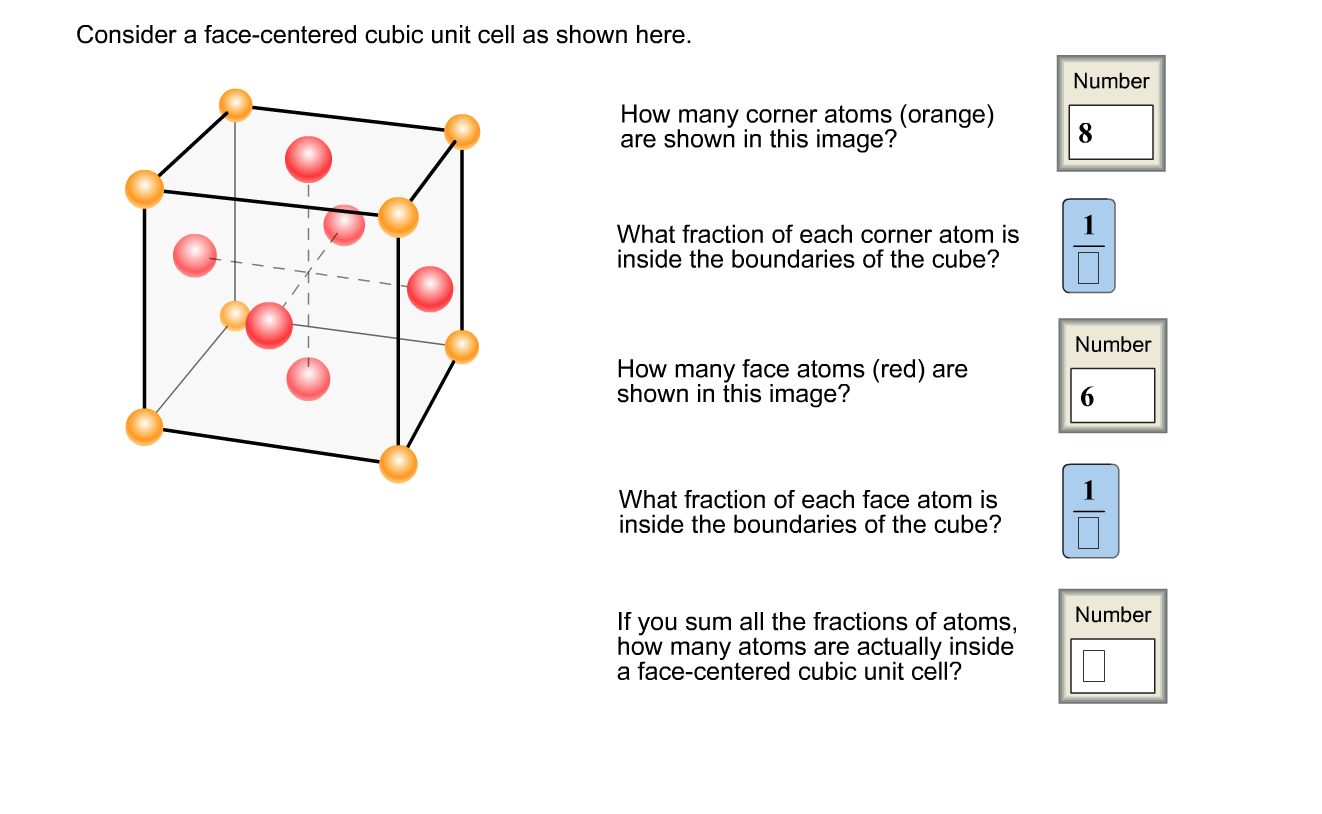
An element has a body-centered cubic (bcc) structure with a cell edge of 288pm. The density...... - YouTube

Silver crystallises in a face - centred cubic in cell. The density of Ag is 10.5 g cm^-3 . Calculate the edge length of the unit cell.

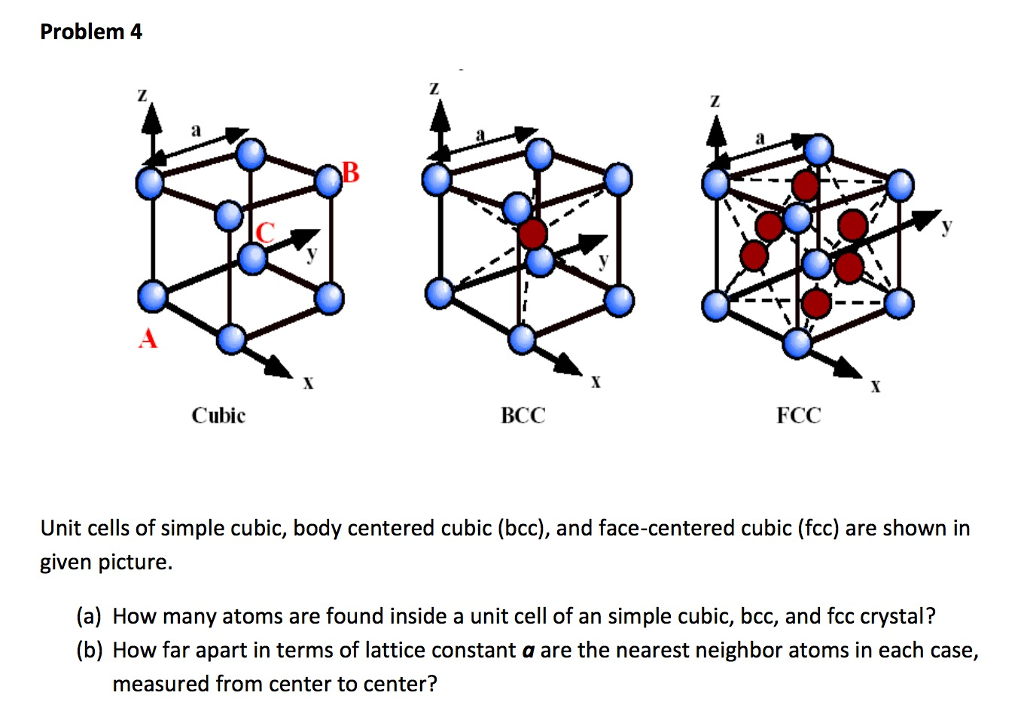
PH0101,UNIT 4,LECTURE 51 PH0101 UNIT 4 LECTURE 5 FACE CENTERED CUBIC STRUCTURE HEXAGONAL CLOSED PACKED STRUCTURE PROBLEMS. - ppt download

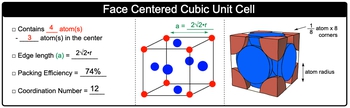
Unit Cell Chemistry, Atomic Radius, Density & Edge Length Calculations, Close Packed Structures - YouTube

3D view of the CFC problem: (a) Typical face centred cubic array (b)... | Download Scientific Diagram

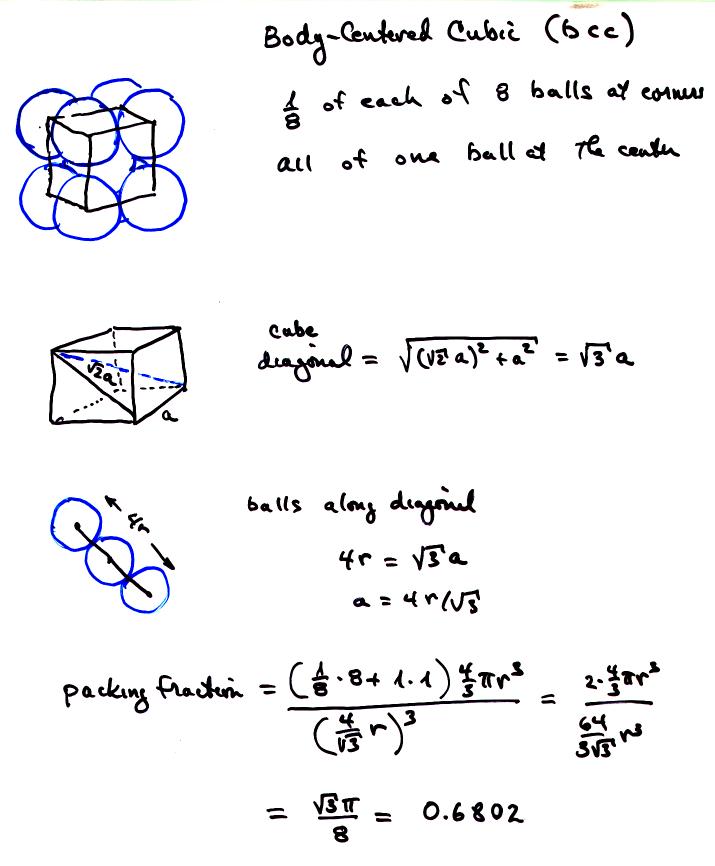
Chemistry - Liquids and Solids (27 of 59) Crystal Structure: Density of the Unit Cell: Body Centered - YouTube



![Solved 3. Face Centered Cubic Structure [10 pts] Platinum is | Chegg.com Solved 3. Face Centered Cubic Structure [10 pts] Platinum is | Chegg.com](https://media.cheggcdn.com/media/1eb/1eb9fa27-1dc7-4453-878c-a7f39a79cbf4/phppeImhN.png)